Global procurement of premium APIs and ingredients for branded and generic formulations.
Ideas & Ingredients
Aligned with Ingenuity, Integrity & Agility
ZUVCHEM supports pharmaceutical, nutraceutical and veterinary organizations by combining high-quality ingredients, regulatory expertise and global sourcing into a single integrated approach.
WHAT WE DO
Integrated solutions

Strategic Sourcing

Custom Development
Tailored ingredient solutions engineered for efficacy, compliance, and competitive success.
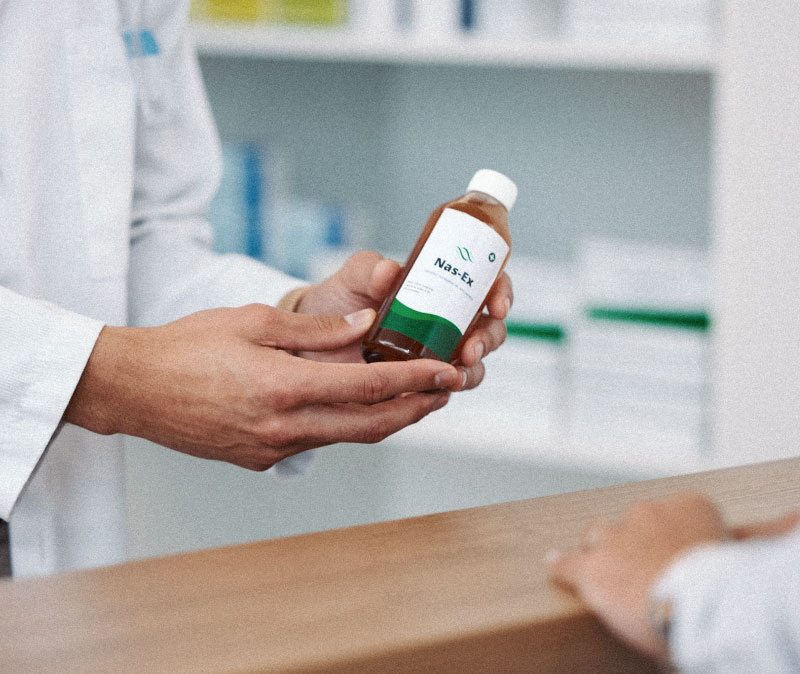
Regulatory Expertise
Seamless support through FDA, EPA, DEA, and USDA frameworks, from registration to approval.

Customer Focused
Client centric solutions backed by a trusted global network of best-in-class manufacturing partners.
Each solution is designed to reduce complexity while maintaining the highest standards of quality and regulatory alignment.
COMPLIANCE MADE SIMPLE
Regulatory expertise without unnecessary complexity
Rather than approaching regulatory activities as isolated tasks, we integrate them directly into sourcing, development, and commercialization strategy. This integrated model enables clients to accelerate timelines, mitigate risk, and maintain full regulatory oversight at every stage.
From FDA registrations to broader U.S. market requirements, our team supports structured, compliant expansion with clarity and control.

WHAT WE DO
Integrated solutions
Each solution is designed to reduce complexity while maintaining the highest standards of quality and regulatory alignment.
COMPLIANCE MADE SIMPLE
Regulatory expertise without unnecessary complexity
Rather than approaching regulatory activities as isolated tasks, we integrate them directly into sourcing, development, and commercialization strategy. This integrated model enables clients to accelerate timelines, mitigate risk, and maintain full regulatory oversight at every stage.
From FDA registrations to broader U.S. market requirements, our team supports structured, compliant expansion with clarity and control.

OUR PRODUCT FOCUS
High-quality ingredients for pharmaceutical innovation
We provide access to a curated portfolio of pharmaceutical and nutraceutical ingredients sourced from qualified, GMP-compliant manufacturers.
Every product is supported by technical documentation, quality systems and regulatory alignment to meet demanding market standards.
Specialty Chemicals & Excipients
Nutraceutical and specialty ingredients
OUR PRODUCT FOCUS
High-quality ingredients for pharmaceutical innovation
We provide access to a curated portfolio of pharmaceutical and nutraceutical ingredients sourced from qualified, GMP-compliant manufacturers.
Every product is supported by technical documentation, quality systems and regulatory alignment to meet demanding market standards.
Active pharmaceutical ingredients (APIs)
Specialty Chemicals
Nutraceutical and specialty ingredients
EXPERTISE THAT DRIVES RESULTS
A team built around
regulated industries

Behind every ZUVCHEM project is a multidisciplinary team with experience in
- Regulatory Affairs
- Pharmaceutical and Nutraceutical Development
- Manufacturing and Quality Systems
- Global Sourcing and Distribution
Rooted in our name, ZUV, meaning life, our mission is to advance health and wellness through science, integrity, and purpose.
With 20+ years of industry expertise, ZUVCHEM stands at the intersection of technical precision, regulatory insight, and global sourcing agility. We empower pharmaceutical, nutraceutical, and veterinary innovators across the U.S. to navigate complex regulatory pathways and bring meaningful products to market efficiently and compliantly.
QUALITY AND COMPLIANCE
Excellence starts with accountability
All products and partners are selected according to strict quality and compliance criteria. We work with FDA-registered and cGMP-certified facilities and apply robust quality control and audit processes.
For us, compliance is not a checkbox. It is the foundation of trust, reliability and long-term collaboration.

NEWS AND INDUSTRY PRESENCE
Active in the
pharmaceutical ecosystem
ZUVCHEM actively participates in industry events and follows market developments across regulated sectors. In our News section you will find


CONTACT US
Ready to move forward
Whether you are developing a new product, entering the U.S. market or optimizing your regulatory strategy, our team is ready to support your next step.
